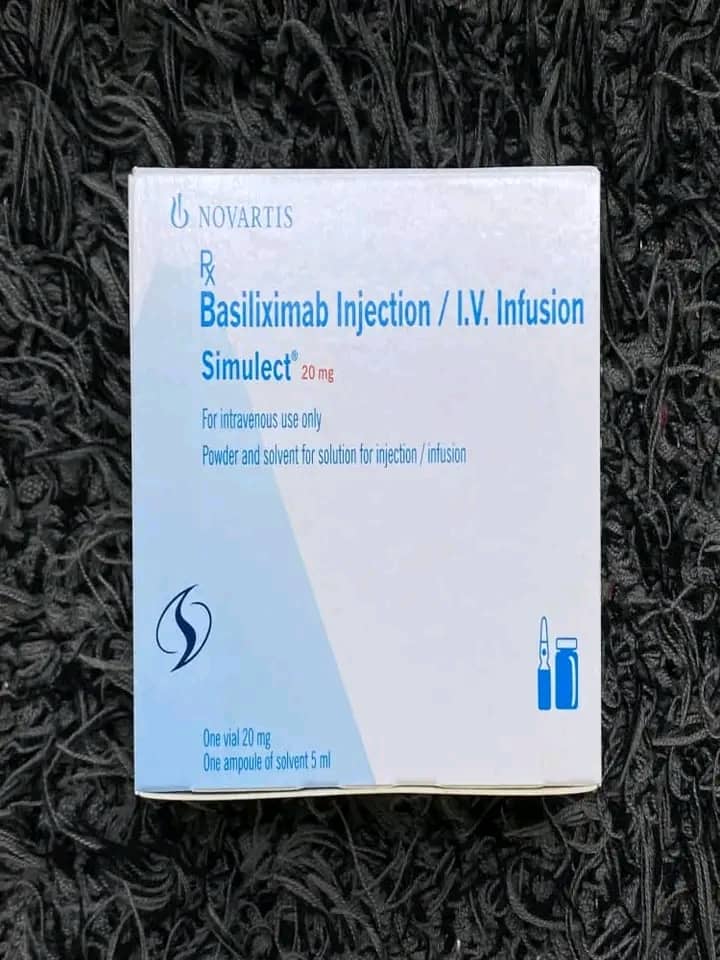
The World Health Organisation (WHO) has issued a warning to countries over the circulation of falsified SIMULECT (basiliximab), an immunosuppressant used to prevent organ rejection in kidney transplant patients.
According to WHO, counterfeit versions of the medicine have been detected in Rwanda, Bulgaria and Türkiye, with the first cases reported in December 2024 and resurfacing in November 2025.
The organisation says the falsified product poses serious health risks as it misrepresents the genuine drug’s identity, composition and source, potentially endangering patients who rely on it for post-transplant treatment.
In response to the global alert, the Pharmacy and Medicines Regulatory Authority (PMRA) has stated that SIMULECT is not registered for use in Malawi and is therefore unlikely to be found on the local market.
PMRA explained that due to its unregistered status, the medicine is not authorised for importation or distribution in the country, reducing the chances of the falsified version reaching Malawian patients.
However, the Authority has urged the public, health professionals, pharmaceutical retailers and importers to remain vigilant amid the international circulation of the counterfeit product.
PMRA is encouraging people to report any suspicious medical products or adverse drug reactions to ensure swift intervention and protect public safety.
The regulator further warned that anyone who comes across the falsified SIMULECT should avoid using it and report it immediately to the nearest health facility or PMRA office.